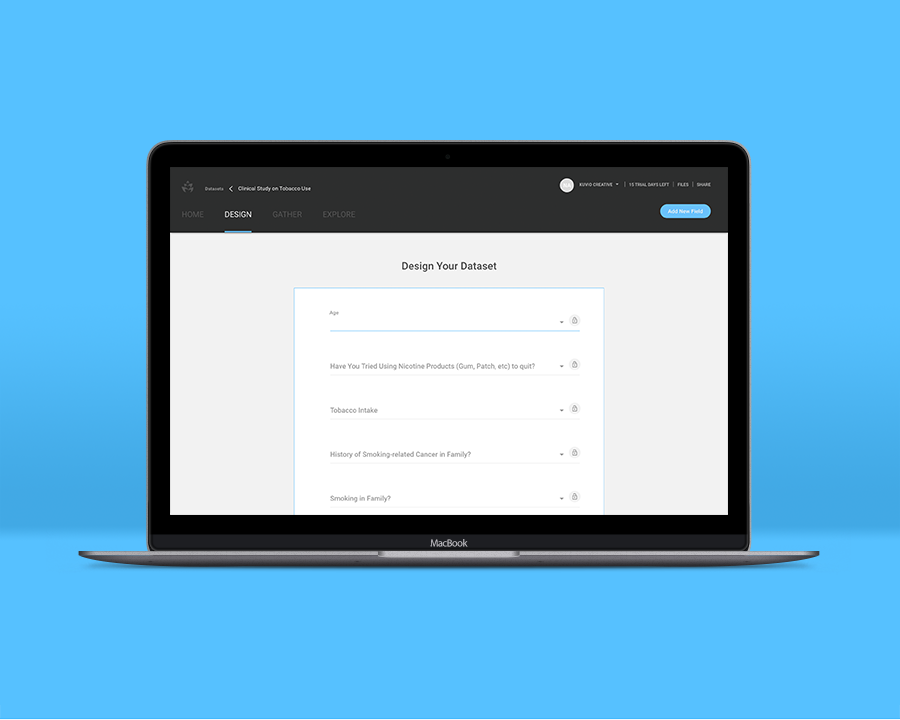
You’re engineering a prescription called Nictophane that will treat common side effects associated with tobacco usage. The results of a previous clinical trial indicate that age and genetics are strongly correlated to the severity of participants’ symptoms, though you will need participants to test the prescription in order to achieve more accurate results. Based on promising preliminary results, you decide to focus on participants between the ages of 40-60 with a genetic history of lung cancer and other tobacco-related ailments.
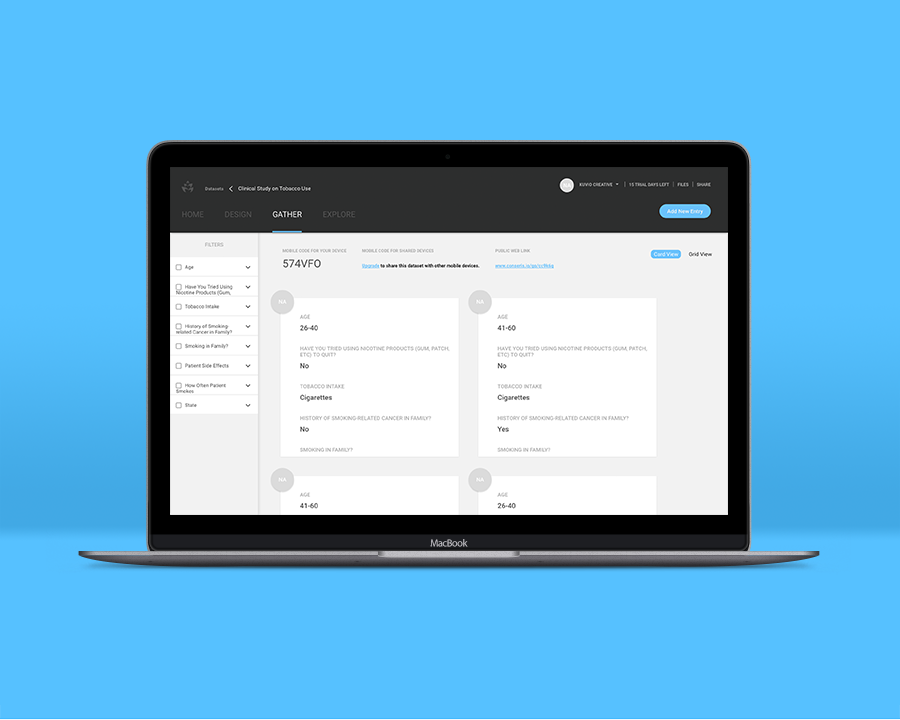
You collect statistics from 10 previous clinical trial participants and ask that they return to test your newly-engineered prescription. You then decide to administer a placebo drug to an additional 10 participants to gauge the effectiveness of your prescription. Since the drug is designed for participants of a specific age and genetic demographic, you use Conseris to hyper-focus your research on these metrics. The customization options within Conseris allow you to view, edit, and change your results under a microscope with fine attention to detail, so you can effectively determine if your prescription has its intended effect on trial participants.
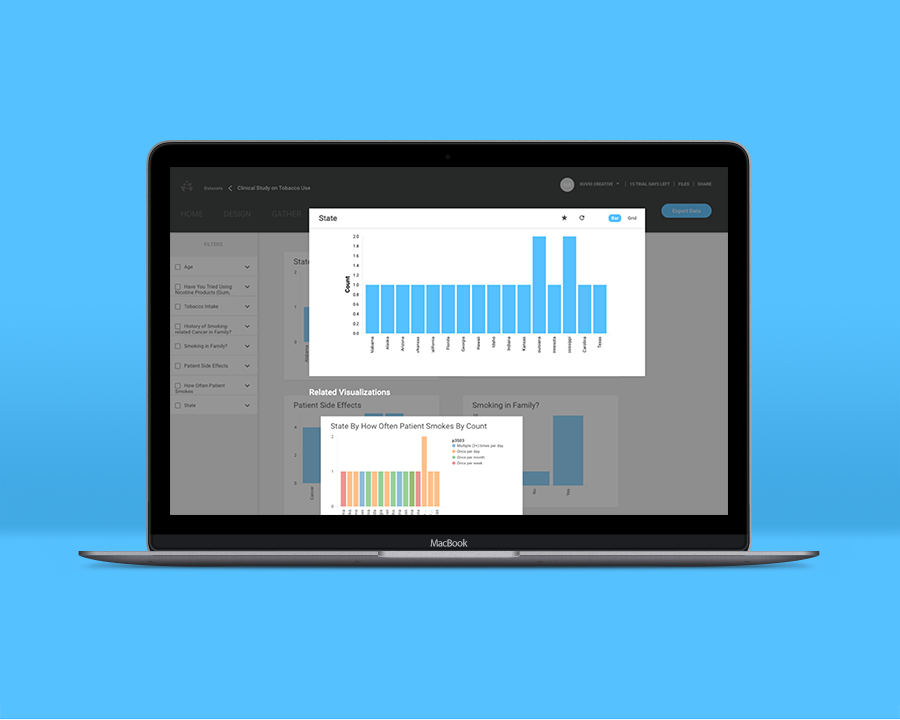
Over the following month, you ask your trial participants to log into Conseris and enter their tobacco-related side effects and symptoms, along with basic information about the severity of these symptoms. The organized form options within the app make logging fast and efficient, streamlining processes that used to require manual entry. The results you glean from your participants indicate that the drug was 70% effective in managing mild tobacco-related symptoms including shortness of breath, coughing, and chest pain.
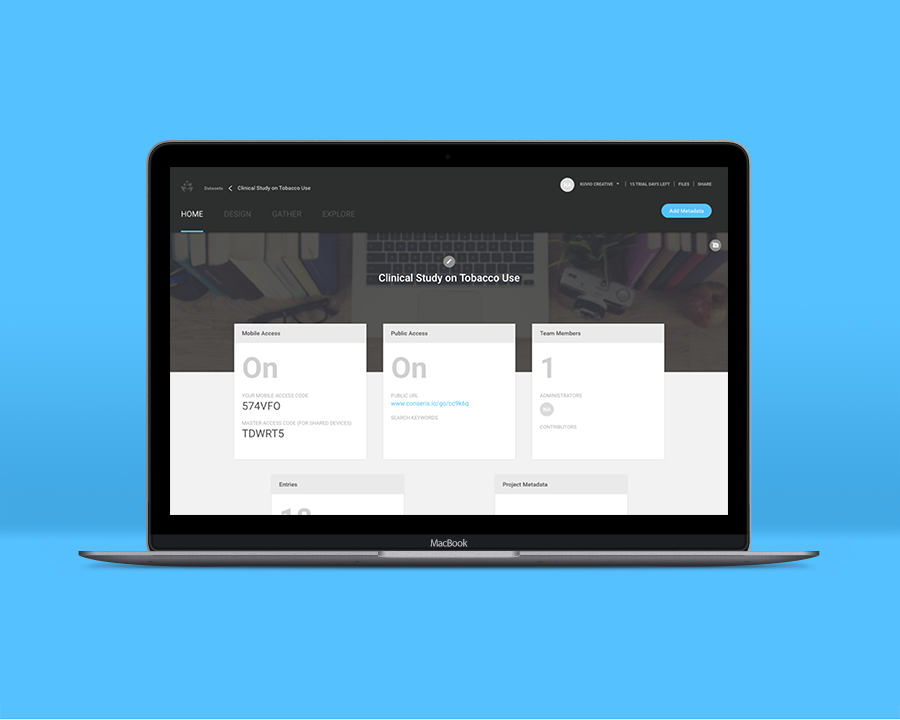
Your results indicated potential in your team’s prescription. The stunning presentation options in Conseris made your trial’s results easy to share and present, and the administrative board agreed to fund additional research on Nictophane.
Eager to learn more about how Conseris can lead to your next biomedical industry breakthrough? Try us out today.